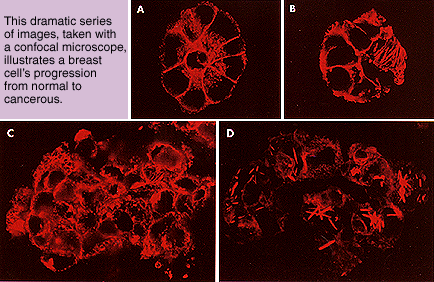
The assay, which was developed in collaboration with Ole Petersen, a
professor of
pathology from Denmark, provides cells in a culture with basement
membrane ECM
and a special synthetic medium. In the presence of this combination, normal
and
cancerous breast cells once again begin to act like normal and cancerous
breast
cells. Within six days, each is readily distinguishable.
The assay, which was developed in collaboration with Ole Petersen, a
professor of
pathology from Denmark, provides cells in a culture with basement
membrane ECM
and a special synthetic medium. In the presence of this combination, normal
and
cancerous breast cells once again begin to act like normal and cancerous
breast
cells. Within six days, each is readily distinguishable."With this assay, we can add certain things to our cells or knock out crucial genes and determine whether the cells become malignant," says Bissell. "We can also replace the genes that tumor cells seem to lack and see whether we can make them normal."
Bissell and a team that included Dr. Patricia Steeg of the National Cancer Institute have already done this with a gene called "nm23." This is considered to be a metastatic tumor suppressor gene, because when it is lost, the cell loses its ability to stay in one place and starts moving throughout the body. Damaged nm23 genes are found in a variety of tumors in addition to breast cancer. The research teams of Bissell and Steeg used the 3-D assay to discover that overexpression of the nm23 gene resulted in the formation of excessive amounts of basement membrane ECM, which in turn inhibited cell growth. The effect of overexpressed nm23 was "dramatic," according to Bissell.
"We found that metastatic cells that overexpress a transfected nm23 gene form small, semi-normal (cell) structures, whereas parent (tumor) cells continue to grow in a disorganized manner. Our data suggest that production of basement membrane initiates a cascade of cellular responses that lead to growth arrest," she says. "This is the first cause and effect data concerning nm23 and mammalian breast cell differentiation."
Bissell and her colleagues now plan to use the assay to study two other tumor suppressor genes, called RB and p53.
The research conducted by Bissell and the many colleagues who have collaborated with her over the past two decades has laid a solid foundation for understanding the physiological basis of breast cancer. Bissell's message-that cells with the same genetic material behave very differently when confronted with different microenvironments-has become a clarion call to cancer researchers everywhere. As for the disease that was for so many years unmentionable, it is now being slowly un- masked. For women in this country and elsewhere, that is good news with no debate.
For women with metastatic breast cancer, few developments in chemotherapy have stirred higher hopes than the recent approval by the Food and Drug Administration of taxol, a drug derived from the bark of the Pacific yew tree. In clinical tests, taxol effectively curtailed the spread of malignancy. The down side was a rise in heart toxicity. Experts say that a safer and even more potent form of taxol could be synthesized if scientists had sufficient knowledge of how the taxol molecule interacts with vital cell proteins to fight cancer. A major step toward this goal was recently taken when scientists at Berkeley Lab produced an atomic-scale, high-resolution three-dimensional image of the site where taxol interacts with its target (right).
This breakthrough arose from on-going research in which Berkeley Lab scientists have been using advanced electron crystallography techniques to produce three-dimensional images of a protein called tubulin. Tubulin is the major constituent of microtubules, which are hollow cylinders-like tiny drinking straws- that serve as part of the skeletal system for cells and are crucial to a number of vital functions, including mitosis (cell division). The Berkeley Lab images revealed the all-important site where taxol binds to tubulin and prevents the protein from performing its necessary tasks.
For a cell to divide, the microtubule skeleton that gives it shape must disassemble, reform into spindles across which duplicate sets of DNA material line up, disassemble once more, and finally reform into skeletal systems for the two new cells. The extreme flexibility of the tubulin protein enables microtubules to shape-shift through these various formations.
"Taxol is a mitotic stabilizer and when it binds to tubulin the protein loses its flexibility. That means the microtubules can no longer disassemble," says Kenneth Downing, who along with Eva Nogales and Sharon Wolf, all biophysicists with Berkeley Lab's Life Sciences Division, led this research. The taxol imaging team also included Israr Khan and Richard Luduena, from the University of Texas in San Antonio.
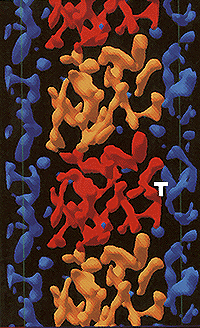 Other studies have shown that taxol binds only to tubulin that has been
polymerized into chains of protofilaments, not to free proteins. The
Berkeley Lab
images indicate that taxol attaches itself at the junction where tubulin
protofilaments are linked, affecting their interaction and locking them into
a
fixed position.
"This is the first time that a taxol binding site has been visualized in
the
tubulin molecule," says Nogales. "Although many questions remain
unanswered, a
number of biological properties can now be interpreted in terms of our
structural
results."
Other studies have shown that taxol binds only to tubulin that has been
polymerized into chains of protofilaments, not to free proteins. The
Berkeley Lab
images indicate that taxol attaches itself at the junction where tubulin
protofilaments are linked, affecting their interaction and locking them into
a
fixed position.
"This is the first time that a taxol binding site has been visualized in
the
tubulin molecule," says Nogales. "Although many questions remain
unanswered, a
number of biological properties can now be interpreted in terms of our
structural
results."
With the latest refinements of their imaging technique and the collection of additional images, the researchers are certain they will be able to construct an atomic model of tubulin and the taxol binding site. Such a model should then enable drug companies to design new and improved versions of taxol.
"We are at the stage now where we feel it is just a matter of collecting and processing enough data to be able to construct an atomic model of the taxol binding site," says Downing.